How To Draw Lewis Structures For Covalent Compounds
10.5: Writing Lewis Structures for Covalent Compounds
- Page ID
- 98061
Learning Objectives
- Depict Lewis structures for covalent compounds.
The following procedure tin be used to construct Lewis electron structures for more complex molecules and ions.
How-to: Constructing Lewis electron structures
i. Make up one's mind the total number of valence electrons in the molecule or ion.
- Add together the valence electrons from each atom. (Recall that the number of valence electrons is indicated by the position of the element in the periodic table.)
- If the species is a polyatomic ion, recall to add or subtract the number of electrons necessary to give the total charge on the ion.
For CO3 2−, for case, we add ii electrons to the total because of the −two charge.
two. Accommodate the atoms to evidence specific connections.
- When there is a central cantlet, it is commonly the least electronegative element in the chemical compound. Chemists ordinarily list this central atom first in the chemical formula (as in CCliv and CO3 two−, which both have C equally the central atom), which is another inkling to the compound's structure.
- Hydrogen and the halogens are almost always connected to only one other atom, and then they are usually concluding rather than central.
3. Place a bonding pair of electrons between each pair of side by side atoms to give a single bond.
- In H2O, for example, there is a bonding pair of electrons betwixt oxygen and each hydrogen.
4. Beginning with the concluding atoms, add enough electrons to each atom to requite each cantlet an octet (two for hydrogen).
- These electrons will usually be lone pairs.
v. If any electrons are left over, place them on the central atom.
- We will explain later that some atoms are able to accommodate more than eight electrons.
half-dozen. If the fundamental atom has fewer electrons than an octet, use lone pairs from last atoms to form multiple (double or triple) bonds to the key atom to accomplish an octet.
- This will not change the number of electrons on the last atoms.
7. Concluding bank check
- Ever make sure all valence electrons are accounted for and that each atom has an octet of electrons, except for hydrogen (with two electrons).
- The fundamental atom is usually the to the lowest degree electronegative chemical element in the molecule or ion; hydrogen and the halogens are usually terminal.
Now let's utilize this procedure to some particular compounds, kickoff with one we have already discussed.
Instance \(\PageIndex{1}\): Water
Write the Lewis Structure for H2O.
Solution
| Steps for Writing Lewis Structures | Case \(\PageIndex{1}\) |
|---|---|
| 1. Determine the full number of valence electrons in the molecule or ion. | Each H atom (group 1) has ane valence electron, and the O atom (grouping 16) has 6 valence electrons, for a full of 8 valence electrons. |
| 2. Arrange the atoms to evidence specific connections. | H O HBecause H atoms are about e'er terminal, the organization inside the molecule must exist HOH. |
| iii. Place a bonding pair of electrons between each pair of adjacent atoms to requite a single bond. four. Beginning with the last atoms, add enough electrons to each cantlet to give each atom an octet (2 for hydrogen). | Placing i bonding pair of electrons between the O atom and each H atom gives H -O- Hwith iv electrons left over. Each H cantlet has a full valence shell of ii electrons. |
| v. If whatever electrons are left over, place them on the cardinal atom. | Adding the remaining 4 electrons to the oxygen (as ii lone pairs) gives the following structure: |
| 6. If the primal atom has fewer electrons than an octet, use lone pairs from concluding atoms to form multiple (double or triple) bonds to the cardinal atom to accomplish an octet. | Not necessary. |
| 7. Final check. | The Lewis structure gives oxygen an octet and each hydrogen 2 electrons. |
Example \(\PageIndex{2}\)
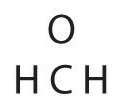
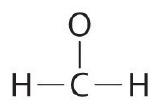
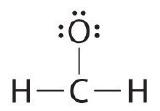
Write the Lewis construction for the \(CH_2O\) molecule
Solution
| Steps for Writing Lewis Structures | Example \(\PageIndex{2}\) |
|---|---|
| ane. Determine the total number of valence electrons in the molecule or ion. | Each hydrogen cantlet (group 1) has one valence electron, carbon (group 14) has 4 valence electrons, and oxygen (group 16) has 6 valence electrons, for a total of [(2)(1) + 4 + vi] = 12 valence electrons. |
| 2. Conform the atoms to bear witness specific connections. | Considering carbon is less electronegative than oxygen and hydrogen is normally terminal, C must exist the primal cantlet. |
| 3. Place a bonding pair of electrons betwixt each pair of adjacent atoms to give a single bond. | Placing a bonding pair of electrons between each pair of bonded atoms gives the following: 6 electrons are used, and six are left over. |
| iv. Beginning with the concluding atoms, add enough electrons to each atom to give each atom an octet (two for hydrogen). | Adding all half dozen remaining electrons to oxygen (as three lone pairs) gives the following: Although oxygen now has an octet and each hydrogen has 2 electrons, carbon has simply 6 electrons. |
| 5. If whatsoever electrons are left over, identify them on the primal atom. | Not necessary. There are no electrons left to place on the central atom. |
| six. If the central atom has fewer electrons than an octet, use solitary pairs from terminal atoms to course multiple (double or triple) bonds to the central atom to accomplish an octet. | To requite carbon an octet of electrons, we use one of the lonely pairs of electrons on oxygen to form a carbon–oxygen double bond: |
| 7. Final cheque | Both the oxygen and the carbon now have an octet of electrons, so this is an acceptable Lewis electron structure. The O has two bonding pairs and two solitary pairs, and C has four bonding pairs. This is the structure of formaldehyde, which is used in embalming fluid. |
Exercise \(\PageIndex{i}\)
Write Lewis electron structures for CO2 and SCltwo, a vile-smelling, unstable red liquid that is used in the industry of rubber.
- Answer COii
-
.


- Answer SCl2
-
.
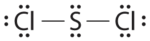
The United States Supreme Courtroom has the unenviable chore of deciding what the law is. This responsibility tin can be a major challenge when at that place is no clear principle involved or where there is a new situation non encountered before. Chemistry faces the same challenge in extending bones concepts to fit a new situation. Drawing of Lewis structures for polyatomic ions uses the same approach, just tweaks the process a little to fit a somewhat dissimilar set of circumstances.
Writing Lewis Structures for Polyatomic Ions
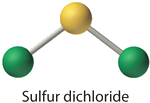
Think that a polyatomic ion is a group of atoms that are covalently bonded together and which acquit an overall electric charge. The ammonium ion, \(\ce{NH_4^+}\), is formed when a hydrogen ion \(\left( \ce{H^+} \right)\) attaches to the lone pair of an ammonia \(\left( \ce{NH_3} \right)\) molecule in a coordinate covalent bond.
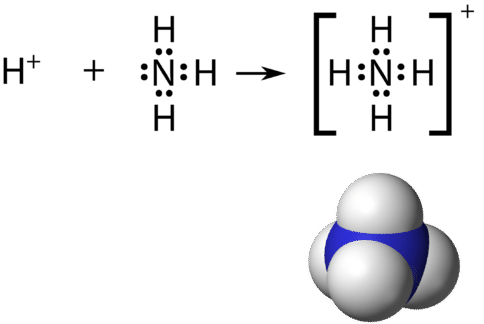
When cartoon the Lewis structure of a polyatomic ion, the charge of the ion is reflected in the number of total valence electrons in the construction. In the case of the ammonium ion:
\(i \: \ce{Northward}\) atom \(= five\) valence electrons
\(4 \: \ce{H}\) atoms \(= 4 \times 1 = iv\) valence electrons
subtract 1 electron for the \(1+\) charge of the ion
total of 8 valence electrons in the ion
Information technology is customary to put the Lewis structure of a polyatomic ion into a large set of brackets, with the charge of the ion as a superscript outside of the brackets.
Practice \(\PageIndex{two}\)
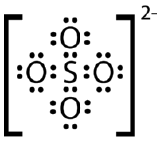
Draw the Lewis electron dot structure for the sulfate ion.
- Answer
-
Exceptions to the Octet Rule
Equally of import and useful as the octet rule is in chemical bonding, in that location are some well-known violations. This does not mean that the octet rule is useless—quite the contrary. As with many rules, there are exceptions, or violations.
There are iii violations to the octet rule. Odd-electron molecules stand for the first violation to the octet rule. Although they are few, some stable compounds have an odd number of electrons in their valence shells. With an odd number of electrons, at least one atom in the molecule will take to violate the octet rule. Examples of stable odd-electron molecules are NO, NO 2 , and ClO ii . The Lewis electron dot diagram for NO is as follows:
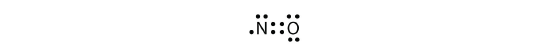
Although the O atom has an octet of electrons, the North atom has only vii electrons in its valence shell. Although NO is a stable chemical compound, it is very chemically reactive, equally are most other odd-electron compounds.
Electron-deficient molecules stand for the second violation to the octet rule. These stable compounds take less than eight electrons around an atom in the molecule. The most common examples are the covalent compounds of beryllium and boron. For instance, beryllium can form 2 covalent bonds, resulting in only four electrons in its valence shell:
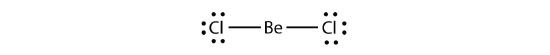
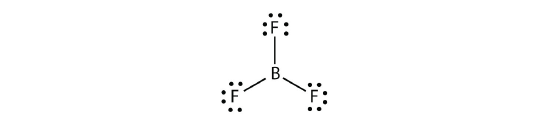
Boron commonly makes only three covalent bonds, resulting in only six valence electrons effectually the B atom. A well-known example is BF 3 :
The third violation to the octet rule is institute in those compounds with more eight electrons assigned to their valence crush. These are called expanded valence shell molecules. Such compounds are formed simply past primal atoms in the tertiary row of the periodic table or beyond that have empty d orbitals in their valence shells that tin can participate in covalent bonding. One such compound is PF v . The only reasonable Lewis electron dot diagram for this compound has the P atom making 5 covalent bonds:
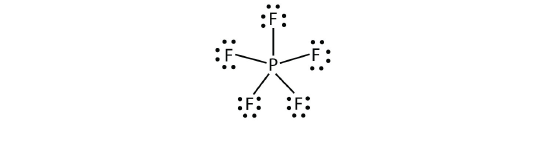
Formally, the P cantlet has ten electrons in its valence vanquish.
Case \(\PageIndex{three}\): Octet Violations
Identify each violation to the octet dominion by cartoon a Lewis electron dot diagram.
- ClO
- SF 6
Solution
a. With one Cl atom and one O cantlet, this molecule has 6 + vii = 13 valence electrons, so it is an odd-electron molecule. A Lewis electron dot diagram for this molecule is as follows:
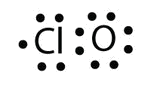
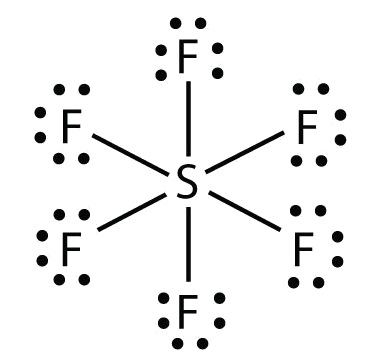
b. In SF half-dozen , the central South atom makes six covalent bonds to the vi surrounding F atoms, then it is an expanded valence shell molecule. Its Lewis electron dot diagram is every bit follows:
Practise \(\PageIndex{iii}\): Xenon Difluoride
Place the violation to the octet dominion in XeF ii by drawing a Lewis electron dot diagram.
- Answer
-
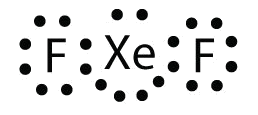
The Xe cantlet has an expanded valence beat out with more than eight electrons around it.
Summary
Lewis dot symbols provide a elementary rationalization of why elements class compounds with the observed stoichiometries. A plot of the overall energy of a covalent bond as a office of internuclear distance is identical to a plot of an ionic pair considering both result from attractive and repulsive forces betwixt charged entities. In Lewis electron structures, we come across bonding pairs, which are shared by two atoms, and lone pairs, which are not shared between atoms. Lewis structures for polyatomic ions follow the aforementioned rules as those for other covalent compounds. There are three violations to the octet rule: odd-electron molecules, electron-deficient molecules, and expanded valence shell molecules.
Source: https://chem.libretexts.org/Courses/College_of_Marin/CHEM_114:_Introductory_Chemistry/10:_Chemical_Bonding/10.05:_Writing_Lewis_Structures_for_Covalent_Compounds
Posted by: hubbardwhatefteld.blogspot.com



0 Response to "How To Draw Lewis Structures For Covalent Compounds"
Post a Comment