How Does Deformation By Drawing Of A Semicrystalline Polymer Affect Its Tensile Strength?
Crystallization of polymers is a process associated with fractional alignment of their molecular bondage. These chains fold together and form ordered regions chosen lamellae, which compose larger spheroidal structures named spherulites.[one] [two] Polymers tin can crystallize upon cooling from melting, mechanical stretching or solvent evaporation. Crystallization affects optical, mechanical, thermal and chemical properties of the polymer. The degree of crystallinity is estimated by different analytical methods and information technology typically ranges between 10 and eighty%, with crystallized polymers often called "semi-crystalline". The backdrop of semi-crystalline polymers are determined not only by the caste of crystallinity, only likewise by the size and orientation of the molecular bondage.
Crystallization mechanisms [edit]

The arrangement of molecular chains in amorphous and semicrystalline polymers.
Solidification from the melt [edit]
Polymers are composed of long molecular chains which grade irregular, entangled coils in the melt. Some polymers retain such a matted structure upon freezing and readily convert into amorphous solids. In other polymers, the chains rearrange upon freezing and form partly ordered regions with a typical size of the social club one micrometer.[3] Although it would be energetically favorable for the polymer chains to marshal parallel, such alignment is hindered by the entanglement. Therefore, inside the ordered regions, the polymer chains are both aligned and folded. Those regions are therefore neither crystalline nor amorphous and are classified equally semicrystalline. Examples of semi-crystalline polymers are linear polyethylene (PE), polyethylene terephthalate (PET), polytetrafluoroethylene (PTFE) or isotactic polypropylene (PP).[4]

The construction of isotactic polypropylene.

The structure of atactic polypropylene.
Whether or not polymers can crystallize depends on their molecular construction – presence of straight chains with regularly spaced side groups facilitates crystallization. For instance, crystallization occurs much easier in isotactic than in the atactic polypropylene form. Atactic polymers crystallize when the side groups are very modest, as in polyvinyl and don't crystallize in case of large substituents like in rubber or silicones.[4]
Nucleation [edit]

Lamellae course during crystallization from the melt. The arrow shows the management of temperature gradient.[5]
Nucleation starts with modest, nanometer-sized areas where as a event of oestrus motility some chains or their segments occur parallel. Those seeds can either dissociate, if thermal movement destroys the molecular order, or grow further, if the grain size exceeds a certain disquisitional value.[4] [v]
Autonomously from the thermal mechanism, nucleation is strongly affected by impurities, dyes, plasticizers, fillers and other additives in the polymer. This is too referred to as heterogeneous nucleation. This issue is poorly understood and irregular, then that the same additive can promote nucleation in i polymer, but not in another. Many of the good nucleating agents are metal salts of organic acids, which themselves are crystalline at the solidification temperature of the polymer solidification.[5]
Crystal growth from the melt [edit]

Schematic model of a spherulite. Blackness arrows indicate management of molecular alignment
Crystal growth is accomplished past the further addition of folded polymer chain segments and only occurs for temperatures below the melting temperature Tm and above the glass transition temperature Tone thousand. Higher temperatures destroy the molecular organisation and beneath the glass transition temperature, the movement of molecular chains is frozen.[half-dozen] Nevertheless, secondary crystallization can keep fifty-fifty below Tg, in the time scale of months and years. This process affects mechanical properties of the polymers and decreases their volume because of a more than compact packing of aligned polymer chains.[4] [7]
The chains interact via various types of the van der Waals forces. The interaction strength depends on the altitude between the parallel chain segments and it determines the mechanical and thermal properties of the polymer.[8]
The growth of the crystalline regions preferably occurs in the management of the largest temperature slope and is suppressed at the peak and lesser of the lamellae past the amorphous folded parts at those surfaces. In the case of a stiff slope, the growth has a unidirectional, dendritic graphic symbol.[nine] However, if temperature distribution is isotropic and static and then lamellae grow radially and form larger quasi-spherical aggregates chosen spherulites. Spherulites have a size between nigh 1 and 100 micrometers[three] and course a large variety of colored patterns (come across, due east.g. front images) when observed between crossed polarizers in an optical microscope, which oftentimes include the "maltese cross" pattern and other polarization phenomena caused by molecular alignment within the individual lamellae of a spherullite.[ii] [4] [5]
Crystallization by stretching [edit]
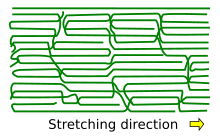
The arrangement of the molecule chains upon crystallization by stretching.[4]
The above mechanism considered crystallization from the melt, which is important for injection molding of plastic components. Another blazon of crystallization occurs upon extrusion used in making fibers and films.
In this process, the polymer is forced through, east.g., a nozzle that creates tensile stress which partially aligns its molecules. Such alignment tin can be considered as crystallization and it affects the material properties. For example, the strength of the fiber is greatly increased in the longitudinal direction, and optical properties show big anisotropy along and perpendicular to the fiber centrality. Such anisotropy is more enhanced in presence of rod-like fillers such as carbon nanotubes, compared to spherical fillers.[10] Polymer strength is increased not only by extrusion, but too past blow molding, which is used in the production of plastic tanks and PET bottles.[8] Some polymers which do not crystallize from the melt, can be partially aligned by stretching.[xi]
Some elastomers which are amorphous in the unstrained state undergo rapid crystallization upon stretching.
Crystallization from solution [edit]
Polymers can likewise be crystallized from a solution or upon evaporation of a solvent. This process depends on the degree of dilution: in dilute solutions, the molecular chains have no connection with each other and be every bit a split polymer coils in the solution. Increase in concentration which can occur via solvent evaporation, induces interaction between molecular bondage and a possible crystallization as in the crystallization from the cook.[12] Crystallization from solution may result in the highest degree of polymer crystallinity. For example, highly linear polyethylene can form platelet-similar single crystals with a thickness on the order 10–twenty nm when crystallized from a dilute solution. The crystal shape can be more complex for other polymers, including hollow pyramids, spirals and multilayer dendritic structures.[2]
A very dissimilar process is atmospheric precipitation; information technology uses a solvent which dissolves individual monomers just not the resulting polymer. When a certain degree of polymerization is reached, the polymerized and partially crystallized product precipitates out of the solution. The charge per unit of crystallization tin can be monitored past a technique which selectively probes the dissolved fraction, such as nuclear magnetic resonance.[12]
Confined crystallization [edit]
When polymers crystallize from an isotropic, majority of melt or concentrated solution, the crystalline lamellae (ten to 20 nm in thickness) are typically organized into a spherulitic morphology every bit illustrated above. However, when polymer chains are bars in a space with dimensions of a few tens of nanometers, comparable to or smaller than the lamellar crystal thickness or the radius of gyration, nucleation and growth tin can exist dramatically afflicted. As an instance, when a polymer crystallizes in a confined ultrathin layer, the isotropic spherulitic organization of lamellar crystals is hampered and solitude can produce unique lamellar crystal orientations.[13] Sometimes the concatenation alignment is parallel to the layer plane and the crystals are organized every bit ''on-edge'' lamellae. In other cases, "in-aeroplane" lamellae with concatenation orientation perpendicular to the layers are observed.
The unique crystal orientation of confined polymers imparts anisotropic properties. In one example the large, in-plane polymer crystals reduce the gas permeability of nanolayered films by almost 2 orders of magnitude.[xiii]
Degree of crystallinity [edit]
The fraction of the ordered molecules in polymer is characterized by the degree of crystallinity, which typically ranges between 10% and lxxx%.[4] College values are only achieved in materials having small-scale molecules, which are commonly brittle, or in samples stored for long time at temperatures just nether the melting betoken. The latter procedure is plush and is practical simply in special cases.
Most methods of evaluating the degree of crystallinity presume a mixture of perfect crystalline and totally disordered areas; the transition areas are expected to amount to several percent. These methods include density measurement, differential scanning calorimetry (DSC), X-ray diffraction (XRD), infrared spectroscopy and nuclear magnetic resonance (NMR). The measured value depends on the method used, which is therefore quoted together with the caste of crystallinity.[4]
In improver to the above integral methods, the distribution of crystalline and baggy regions can be visualized with microscopic techniques, such every bit polarized light microscopy and transmission electron microscopy.
- Density measurements
- Crystalline areas are mostly more densely packed than baggy areas. This results in a college density, up to fifteen% depending on the material. For instance, polyamide half-dozen (nylon) has crystalline density ρc = 1.24 yard/cm3 and amorphous density ρa = 1.08 g/cmiii). However, wet which is often nowadays in the sample does affect this blazon of measurement.[half-dozen]
- Calorimetry
- Additional energy is released upon melting a semicrystalline polymer. This energy can exist measured with differential scanning calorimetry and compared with that released upon melting of the standard sample of the same material with known crystallization degree.[half dozen] [14]
- 10-ray diffraction
- Regular arrangement of atoms and molecules produce sharp diffraction peaks whereas amorphous regions issue in wide halos. The diffraction pattern of polymers usually contains a combination of both. Degree of crystallinity can be estimated by integrating the relative intensities of the peaks and halos.[6]
- Infrared spectroscopy (IR)
- Infrared assimilation or reflection spectra from crystalline polymers comprise additional peaks which are absent-minded in baggy materials with the same composition. These signals may originate from deformation vibrations of the regular arrangement of molecular bondage. From the assay of these bands, the caste of crystallinity can be estimated.[6]
- Nuclear magnetic resonance (NMR)
- crystalline and amorphous areas differ by the mobility of protons. The latter can be monitored through the line shape of NMR signals and used to estimate the degree of crystallinity.[6]
Kinetics of polymer crystallization [edit]
The methods used to decide the degree of crystallinity can be incorporated over time to measure the kinetics of crystallization. The near basic model for polymer crystallization kinetics comes from Hoffman nucleation theory. The crystallization procedure of polymers does not always obey elementary chemical rate equations. Polymers can crystallize through a diversity of different regimes and unlike simple molecules, the polymer crystal lamellae accept two very different surfaces. The two well-nigh prominent theories in polymer crystallization kinetics are the Avrami equation and Lauritzen-Hoffman Growth Theory.[15]
Backdrop of semicrystalline polymers [edit]
Thermal and mechanical properties [edit]
Beneath their drinking glass transition temperature, baggy polymers are unremarkably hard and breakable because of the low mobility of their molecules. Increasing the temperature induces molecular movement resulting in the typical rubber-elastic properties. A abiding force applied to a polymer at temperatures above Tgrand results in a viscoelastic deformation, i.e., the polymer begins to pitter-patter. Estrus resistance is usually given for amorphous polymers simply below the drinking glass transition temperature.[16]
Relatively strong intermolecular forces in semicrystalline polymers prevent softening fifty-fifty in a higher place the glass transition temperature. Their elastic modulus changes significantly only at high (melting) temperature.[4] It also depends on the degree of crystallinity: higher crystallinity results in a harder and more thermally stable, just as well more than breakable fabric, whereas the amorphous regions provide certain elasticity and impact resistance.[2] [5] Some other characteristic feature of semicrystalline polymers is stiff anisotropy of their mechanical properties along the direction of molecular alignment and perpendicular to it.[17]
In a higher place the glass transition temperature amorphous chains in a semi-crystalline polymer are ductile and are able to deform plastically. Crystalline regions of the polymer are linked by the amorphous regions. Necktie molecules forestall the amorphous and crystalline phases from separating under an practical load. When a tensile stress is applied the semi-crystalline polymer commencement deforms elastically. While the crystalline regions remain unaffected past the practical stress, the molecular chains of the amorphous phase stretch. Then yielding, which signifies the onset of plastic deformation of the crystalline regions, occurs.[18]
The molecular mechanism for semi-crystalline yielding involves the deformation of crystalline regions of the fabric via dislocation motility. Dislocations issue in coarse or fine slips in the polymer and pb to crystalline fragmentation and yielding.[19] Fine slip is defined as a modest amount of skid occurring on a big number of planes. Conversely, fibroid slip is a large corporeality of slip on few planes. The yield stress is determined by the cosmos of dislocations and their resistance to motion.[twenty]
Afterward yielding, a neck is formed in the amorphous region and propagates downward the sample length. During necking, the disordered chains align along the tensile direction, forming an ordered structure that demonstrates strengthening due to the molecular reorientation. The flow stress now increases significantly following cervix propagation. Mechanical anisotropy increases and the elastic modulus varies along unlike directions, with a loftier modulus observed in the draw direction. Drawn semi-crystalline polymers are the strongest polymeric materials due to the stress-induced ordering of the molecular chains.[21]
Other defects, such every bit voids, occur in the semi-crystalline polymer nether tensile stress and can drive the formation of the neck. The voids can be observed via small angle 10-ray handful. Different crazes these voids practise not transfer stresses.. Notably, cavitation is non observed under compressive stress or shearing. Evidence suggests that cavitation also impacts the onset of yielding. The voids are associated with the breaking of the baggy phase. The strength of the crystalline phase determines the importance of cavitation in yielding. If the crystalline structures are weak, they deform easily resulting in yielding. Semi-crystalline polymers with strong crystalline regions resist deformation and cavitation, the formation of voids in the amorphous phase, drives yielding.[19]
Equally done in crystalline materials, particles can exist added to semi-crystalline polymers to change the mechanical properties. In crystalline materials the addition of particles works to impede dislocation motion and strengthen the fabric. Notwithstanding, for many semi-crystalline polymers particle fillers weaken the fabric. It has been suggested that for particles to accept a toughening effect in polymers the interparticle matrix ligament thickness must exist smaller than a certain threshold. Crystalline polymers polypropylene and polyethylene display particle strengthening.[22]
Plastics are viscoelastic materials pregnant that under practical stress, their deformation increases with time (pitter-patter). The elastic backdrop of plastics are therefore distinguished co-ordinate to the time scale of the testing to short-time behavior (such every bit tensile examination which lasts minutes), shock loading, the behavior under long-term and static loading, also as the vibration-induced stress.[23]
Optical properties [edit]
Crystalline polymers are unremarkably opaque because of light scattering on the numerous boundaries between the crystalline and amorphous regions. The density of such boundaries is lower and consequentially, the transparency is higher - either for low (amorphous polymer) or loftier (crystalline) degree of crystallinity.[5] For instance, atactic polypropylene is commonly baggy and transparent while syndiotactic polypropylene, which has crystallinity ~fifty%, is opaque.[24] Crystallinity too affects dyeing of polymers: crystalline polymers are more difficult to stain than baggy ones because the dye molecules penetrate through amorphous regions with greater ease.[25]
Meet also [edit]
- Liquid crystal polymer
- Modeling of polymer crystals
References [edit]
- ^ Andrew Keller (1952). "Morphology of crystallizing polymers". Nature. 169 (4309): 913–914. Bibcode:1952Natur.169..913K. doi:10.1038/169913a0. S2CID 4255757.
- ^ a b c d Charles E. Carraher; Raymond Bridegroom Seymour (2003). Seymour/Carraher's polymer chemical science. CRC Printing. pp. 43–45. ISBN978-0-8247-0806-1.
- ^ a b Linda C. Sawyer; David T. Grubb; Gregory F. Meyers (2008). Polymer microscopy. Springer. p. 5. ISBN978-0-387-72627-4.
- ^ a b c d e f yard h i j Chiliad. Due west. Ehrenstein; Richard P. Theriault (2001). Polymeric materials: construction, backdrop, applications. Hanser Verlag. pp. 67–78. ISBN978-ane-56990-310-0.
- ^ a b c d due east f Georg Menges, Edmund Haberstroh, Walter Michaeli, Ernst Schmachtenberg: Plastics Materials Science Hanser Verlag, 2002, ISBN 3-446-21257-4
- ^ a b c d e f GW Becker, Ludwig Bottenbruch, Rudolf Binsack, D. Braun: Engineering Thermoplastics. Polyamides. (in German) Hanser Verlag, 1998 ISBN 3-446-16486-3
- ^ Wilbrand Woebcken, Klaus Stöckhert, HBP Gupta: Plastics Encyclopedia. (in German) Hanser Verlag, 1998, ISBN 3-446-17969-0
- ^ a b Wolfgang Weissbach: Materials science and materials testing. Vieweg + Teubner Verlag, 2007, ISBN three-8348-0295-half dozen
- ^ Dendrite in the IWF Knowledge and Media gGmbH (videos and articles on the dendritic crystallization of polypropylene)
- ^ Patil, Northward; Balzano, L; Portale, G; Rastogi, S (July 2022). "A Study on the Chain−Particle Interaction and Aspect Ratio of Nanoparticles on Structure Evolution of a Linear Polymer". Macromolecules. 43 (xvi): 6749–6759. Bibcode:2010MaMol..43.6749P. doi:ten.1021/ma100636v. ISSN 0024-9297.
- ^ Michael Thielen, Klaus Hartwig, Peter Gust: Blow molding of plastic articles Hanser Verlag, 2006, ISBN 3-446-22671-0
- ^ a b J. Lehmann (1966). "The observation of the crystallization of high polymer substances from the solution by nuclear magnetic resonance". Colloid & Polymer Science. 212 (two): 167–168. doi:10.1007/BF01553085. S2CID 96640893.
- ^ a b Wang, Haopeng; Jong Thou. Keum; Anne Hiltner; Eric Baer; Benny Freeman; Artur Rozanski; Andrzej Galeski (6 February 2009). "Confined Crystallization of Polyethylene Oxide in Nanolayer Assemblies". Scientific discipline. 323 (5915): 757–760. Bibcode:2009Sci...323..757W. doi:x.1126/scientific discipline.1164601. PMID 19197057. S2CID 19276.
- ^ Gottfried W. Ehrenstein, Gabriela Riedel, Pia Trawiel: Practise of thermal analysis of plastics. Hanser Verlag, 2003, ISBN 3-446-22340-1
- ^ Paul C. Painter; Michael G. Coleman (1997). "viii". Fundamentals of Polymer Science An Introductory Text, Second Edition. CRC Printing.
- ^ Joachim Nentwig: Plastic films (in German) Hanser Verlag, 2006, ISBN iii-446-40390-6
- ^ Martin Bonnet: Plastics in engineering applications: properties, processing and applied utilise of polymeric materials. (in German language) Vieweg+Teubner Verlag, 2008 ISBN three-8348-0349-ix
- ^ Courtney, T. H. "Mechanical Beliefs of Materials". Waveland Press (2005) , 392-396
- ^ a b Pawlak, A., Galeski A,. Rozanski, A. Cavitation during deformation of semicrystalline polymers. Progress in Polymer Science. (2014). 921-958
- ^ Bowden, P.B., Young, R.J. Deformation Mechanisms in Crystalline Polymers. Periodical of Materials Scientific discipline. (1974), 2034-2051.
- ^ Courtney, T. H.. Mechanical Behavior of Materials. Waveland Press (2005) , 392-396
- ^ Bartczak, Z., Argon A.S., Weinberg, M. Toughness mechanism in semi-crystalline polymer blends: II. High-density polyethylene toughened with calcium carbonate filler particles. Polymer, 1999. 2347-2365.
- ^ James F. Shackelford (2009). Introduction to Materials Science for Engineers. Prentice Hall. pp. 168–169. ISBN978-0-13-601260-three.
- ^ Andrew J. Peacock; Allison R. Calhoun (2006). Polymer chemical science: backdrop and applications. Hanser Verlag. pp. 286–287. ISBN978-1-56990-397-1.
- ^ Ágnes Tímár-Balázsy; Dinah Eastop (1998). Chemical principles of textile conservation. Butterworth-Heinemann. p. 11. ISBN978-0-7506-2620-0.
Source: https://en.wikipedia.org/wiki/Crystallization_of_polymers
Posted by: hubbardwhatefteld.blogspot.com


0 Response to "How Does Deformation By Drawing Of A Semicrystalline Polymer Affect Its Tensile Strength?"
Post a Comment